NEW YORK, June 20, 2024 /PRNewswire/ -- The
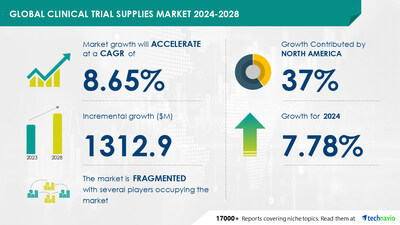
global clinical trial supplies market size is estimated
to grow by USD 1.31 billion from
2024-2028, according to Technavio. The market is estimated to grow
at a CAGR of over 8.65% during the forecast
period. Growth of biopharmaceutical industry is
driving market growth, with a trend towards growing
demand for CROS. However, high cost of clinical trials poses a
challenge. Key market players include Ancillare LP, Avantor Inc.,
Bionical Emas Group, Bushu Pharmaceuticals Ltd., Catalent Inc.,
Clinical Services International Ltd., Clinigen Group Plc, Deutsche
Bahn AG, Emsere B.V, GNH India Pharmaceuticals Ltd., International
Bonded Couriers Inc., Nuvisan GmbH, Obvio Health USA Inc., Parexel International Corp., PCI
Pharma Services, PHOENIX
Pharmahandel GmbH and Co KG, Piramal Enterprises Ltd., Precision
Medicine Group LLC, Thermo Fisher Scientific Inc., and Vertical
Pharma Resources Ltd..
Get a detailed analysis on regions, market
segments, customer landscape, and companies - Click for the
snapshot of this report
|
Forecast
period
|
2024-2028
|
|
Base Year
|
2023
|
|
Historic
Data
|
2018 - 2022
|
|
Segment
Covered
|
End-user
(Pharmaceutical industry, Biologics
industry, and Medical device industry), Type
(Phase III, Phase II, Phase IV and BE/BA studies,
and Phase I), and Geography (North America,
Europe, Asia, and Rest of World (ROW))
|
|
Region
Covered
|
North America, Europe,
Asia, and Rest of World
(ROW)
|
|
Key companies
profiled
|
Ancillare LP, Avantor
Inc., Bionical Emas Group,
Bushu Pharmaceuticals Ltd., Catalent Inc.,
Clinical Services International Ltd., Clinigen Group Plc,
Deutsche Bahn AG, Emsere B.V, GNH India
Pharmaceuticals Ltd., International Bonded
Couriers Inc., Nuvisan GmbH, Obvio Health USA
Inc., Parexel International Corp., PCI Pharma
Services, PHOENIX Pharmahandel GmbH and Co
KG, Piramal Enterprises Ltd., Precision Medicine
Group LLC, Thermo Fisher Scientific Inc., and
Vertical Pharma Resources Ltd.
|
Key Market Trends Fueling Growth
The clinical trial supplies market is experiencing significant
growth due to the increasing outsourcing of clinical trials by
pharmaceutical companies to Contract Research Organizations (CROs).
This trend is driven by the need to optimize operational costs and
focus on research and development. Developed countries are
outsourcing research and testing to emerging economies like
India and Brazil to reduce costs. Key factors fueling
market growth include high R&D spending, increasing disease
prevalence, and a focus on rare diseases. Strategic partnerships
between sponsors and CROs, as well as the decreased risk of
clinical trial failure, further contribute to market
expansion.
The Clinical Trial Supplies market is experiencing significant
growth, driven by the increasing number of drugs in development and
the need for high-quality supplies. Drugs and biologics, clinical
trial procedures, and manufacturing processes are key areas of
focus. The use of technology, such as electronic data capture and
real-time monitoring, is also trending. Cardiovascular, oncology,
and neurology are leading therapeutic areas. Companies are
investing in capacity expansion and strategic collaborations to
meet demand. The market is competitive, with major players
including CROs, CDMOs, and suppliers. Regulatory compliance and
cost containment are ongoing challenges. Overall, the Clinical
Trial Supplies market is dynamic and evolving to meet the demands
of drug development.
Research report provides comprehensive data on
impact of trend. For more details- Download a Sample
Report
Market Challenges
- The clinical trials landscape presents significant challenges,
including patient recruitment and retention, and the need for
extensive safety data collection. These factors contribute to the
rising costs of clinical trials, particularly for drugs treating
chronic diseases. The emergence of novel conjugate vaccines
requires substantial investments in development and manufacturing,
further increasing costs. Phase I clinical trials for innovative
products can cost up to USD60
million. These expenses may impact the growth of the
clinical trial supplies market. To mitigate costs, increasing
patient awareness and education is recommended.
- The Clinical Trial Supplies market faces several challenges in
the drug development process. The complexity of clinical trials
requires a large quantity of supplies, including placebos, active
pharmaceutical ingredients, and packaging materials. The
distribution of these supplies must be efficient and secure to
ensure the integrity of the trial. However, logistical issues such
as temperature control, transportation, and regulatory compliance
can pose significant challenges. Additionally, the cost of clinical
trials continues to rise, putting pressure on suppliers to provide
cost-effective solutions without compromising quality. The use of
technology, such as electronic data capture and real-time inventory
management, can help address some of these challenges. Overall, the
Clinical Trial Supplies market requires a robust and flexible
supply chain to meet the demands of drug development while ensuring
regulatory compliance and maintaining the highest standards of
quality.
For more insights on driver and
challenges - Download a Sample Report
Segment Overview
This clinical trial supplies market report extensively covers
market segmentation by
- End-user
- 1.1 Pharmaceutical industry
- 1.2 Biologics industry
- 1.3 Medical device industry
- Type
- 2.1 Phase III
- 2.2 Phase II
- 2.3 Phase IV and BE/BA studies
- 2.4 Phase I
- Geography
- 3.1 North America
- 3.2 Europe
- 3.3 Asia
- 3.4 Rest of World (ROW)
1.1 Pharmaceutical industry- The Clinical Trial
Supplies Market is a significant sector, supplying essential
materials for pharmaceutical and biotech companies. These supplies
include trial kits, packaging materials, labels, and ancillary
supplies. The market's growth is driven by increasing research and
development activities, regulatory compliance, and technological
advancements. Companies focus on improving efficiency, reducing
costs, and ensuring quality to meet the demands of this market.
For more information on market segmentation with geographical
analysis including forecast (2024-2028) and historic data (2018 -
2022) - Download a Sample Report
Research Analysis
The Clinical Trial Supplies Market encompasses a broad spectrum
of products and services essential for conducting clinical trials.
Key stakeholders, including pharmaceutical and biotech companies,
regulatory bodies, and clinical research organizations (CROs), play
a pivotal role in driving market growth. Buying behavior is
influenced by the prevalence of diseases under investigation,
regulatory support, and industrial collaborations. Drug development
involves various stages, including registered clinical trials,
clinical research activities, and drug approval. Biologics &
biosimilar drugs, oncology, cardiovascular disease, and medical
devices are primary therapeutic areas. Biological drugs,
pharmaceutical drugs, and cold chain distribution are significant
market segments. New drugs and follow-up rates are crucial
performance indicators. Biologics, including monoclonal antibodies
and vaccines, are a significant focus due to their potential in
treating complex diseases like cancer.
Market Research Overview
The Clinical Trial Supplies Market encompasses a range of
products and services essential for the successful conduct of
clinical trials. These include drugs, biologics, placebos, and
clinical trial kits. Drugs and biologics are integral components of
clinical trials, serving as investigational products for testing
and evaluation. Clinical trial kits consist of a collection of
supplies required for trial sites, such as syringes, needles, and
patient diaries. Additionally, services like packaging, labeling,
and distribution of clinical trial supplies are crucial in ensuring
trial efficiency and accuracy. The global Clinical Trial Supplies
Market is driven by the increasing number of clinical trials,
regulatory requirements, and advancements in biotechnology and
pharmaceuticals. The market is expected to grow significantly in
the coming years, with a focus on improving trial quality, reducing
costs, and enhancing patient safety.
Table of Contents:
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation
- End-user
-
- Pharmaceutical Industry
- Biologics Industry
- Medical Device Industry
- Type
-
- Phase III
- Phase II
- Phase IV And BE/BA Studies
- Phase I
- Geography
-
- North America
- Europe
- Asia
- Rest Of World (ROW)
7 Customer Landscape
8 Geographic Landscape
9 Drivers, Challenges, and Trends
10 Company Landscape
11 Company Analysis
12 Appendix
About Technavio
Technavio is a leading global technology research and advisory
company. Their research and analysis focuses on emerging market
trends and provides actionable insights to help businesses identify
market opportunities and develop effective strategies to optimize
their market positions.
With over 500 specialized analysts, Technavio's report library
consists of more than 17,000 reports and counting, covering 800
technologies, spanning across 50 countries. Their client base
consists of enterprises of all sizes, including more than 100
Fortune 500 companies. This growing client base relies on
Technavio's comprehensive coverage, extensive research, and
actionable market insights to identify opportunities in existing
and potential markets and assess their competitive positions within
changing market scenarios.
Contacts
Technavio Research
Jesse Maida
Media & Marketing Executive
US: +1 844 364 1100
UK: +44 203 893 3200
Email: media@technavio.com
Website: www.technavio.com/
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/clinical-trial-supplies-market-size-is-set-to-grow-by-usd-1-31-billion-from-2024-2028--growth-of-biopharmaceutical-industry-to-boost-the-market-growth-technavio-302177062.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/clinical-trial-supplies-market-size-is-set-to-grow-by-usd-1-31-billion-from-2024-2028--growth-of-biopharmaceutical-industry-to-boost-the-market-growth-technavio-302177062.html
SOURCE Technavio